Overview: Vivitra 150 mg Injection contains Trastuzumab (150 mg) and is prescribed as a monoclonal antibody targeted anticancer therapy for the treatment of HER2-positive breast cancer and metastatic gastric cancer by binding to HER2 receptors on tumor cells and slowing their growth. Key Benefits: – Targets HER2 receptors to slow cancer progression – Effective in HER2-positive breast and gastric cancers – Administered by infusion in clinical settings Composition: Trastuzumab (150 mg) How It Works: Trastuzumab is a humanized monoclonal antibody that binds to the human epidermal growth factor receptor 2 (HER2), blocking signaling pathways involved in cancer cell proliferation and survival, leading to reduced tumor growth. Usage Instructions: – Administer only under the supervision of qualified healthcare professionals via intravenous infusion – Follow the prescribed dosage schedule exactly – Not for self-administration Side Effects: – Infusion-related reactions including fever, chills, and nausea – Headache, fatigue, diarrhea – Rare cardiac dysfunction including reduced heart function Precautions: – Monitor cardiac function before and during treatment due to potential cardiotoxicity – Not recommended during pregnancy or breastfeeding unless clearly advised by a physician – Inform your doctor of all current medications prior to infusion Storage Instructions: – Store refrigerated between 2-8°C, protect from light, do not freeze Manufacturer: Generic Pharmaceuticals Disclaimer: To be sold by retail on the prescription of a registered medical practitioner only.
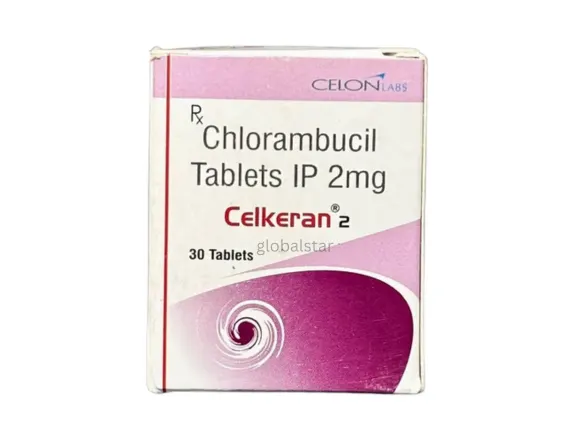
Chlorambucil (2 mg)
Anti Cancer Medicines
Chlorambucil (2 mg)
Vivitra 150 mg Injection contains Trastuzumab (150 mg) and is used as a monoclonal antibody targeted therapy for the treatment of HER2-positive breast cancer and metastatic gastric cancer by binding to HER2 receptors and slowing cancer growth.
Form: Injection Lyophilized Powder
Manufacturer: Generic Pharmaceuticals
Country of Origin: India
| Composition | Trastuzumab (150 mg) |
|---|---|
| Pack Size | 1 Vial |
| Form | Injection Lyophilized Powder |
| Manufacturer | Generic Pharmaceuticals |
| Brand | Vivitra |
| Country of Origin | India |
| Strength | 150 mg |
| Usage Category | Targeted Anticancer / HER2-Positive Cancer Medicines |
| Storage Conditions | do not freeze, protect from light, Store refrigerated between 2-8°C |
| Prescription Required | Yes |
| Administration Route | Intravenous Infusion |
| Dosage Form | Injection Lyophilized Powder |
| Marketed By | Generic Pharmaceuticals |



Reviews
There are no reviews yet.