Description Code
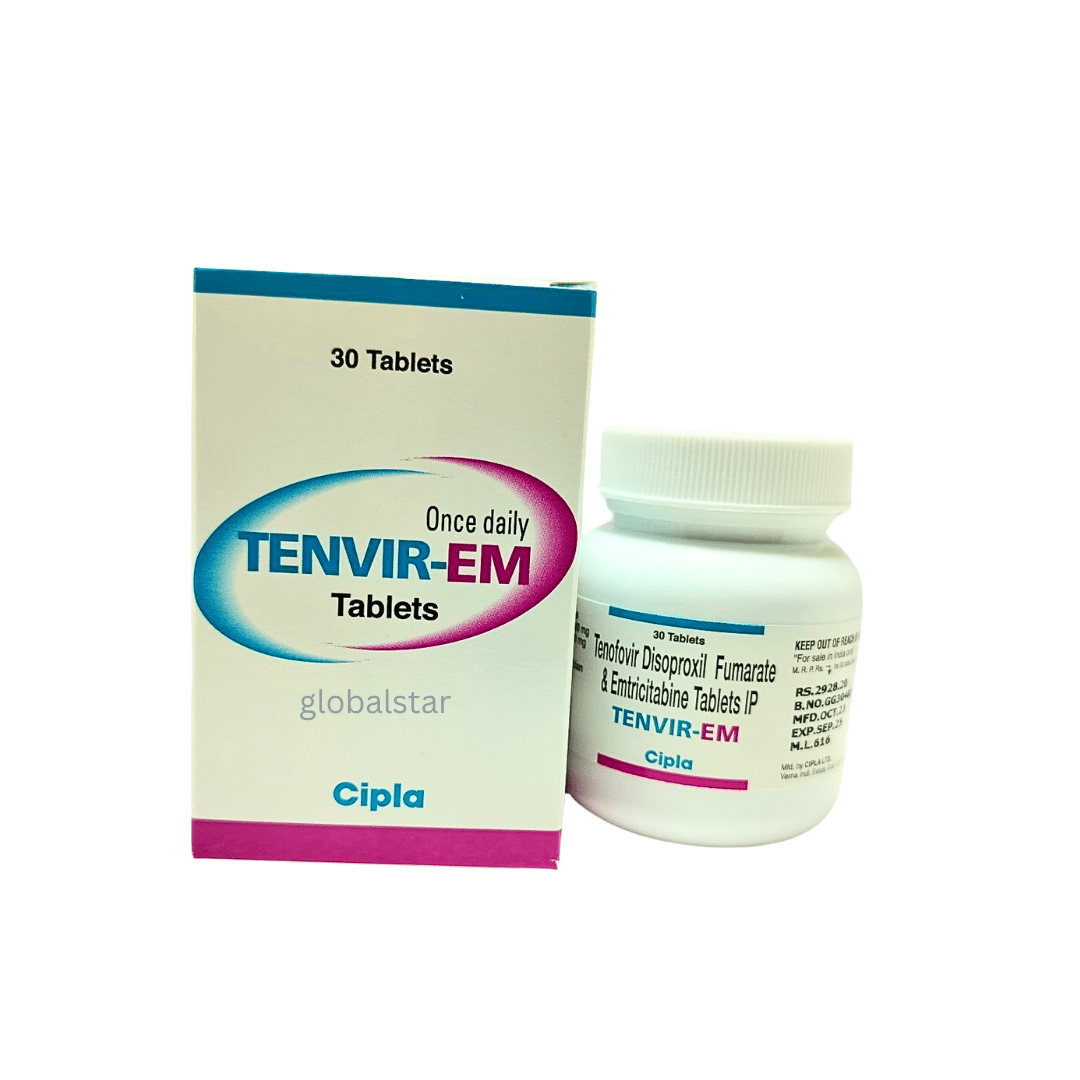
Tenofovir DF 300 mg + Emtricitabine 200 mg
Tenofovir DF 300 mg + Emtricitabine 200 mg
Oral fixed-dose combination of Tenofovir Disoproxil Fumarate 300 mg & Emtricitabine 200 mg for HIV-1 suppression and prophylaxis.
Form: Film-coated tablet
Manufacturer: Cipla Ltd.
Country of Origin: India
| Composition | Tenofovir DF 300 mg + Emtricitabine 200 mg |
|---|---|
| Pack Size | Strip of 30 tablets |
| Form | Film-coated tablet |
| Administration Route | Oral – swallow with water |
| Dosage Regimen | as prescribed, One tablet once daily |
| Usage Indications | HIV-1 treatment & PrEP (Pre-exposure prophylaxis) |
| Country of Origin | India |
| Manufacturer | Cipla Ltd. |
| Prescription Status | Rx required |
| Storage Conditions | protect from moisture, Store below 25 °C |
| Shelf Life | 24 months (typical) |
| Common Side Effects | abdominal pain, abnormal dreams, depression, diarrhea, dizziness, fatigue, headache, insomnia, nausea, rash |
| Precautions | monitor kidney function & bone health; unsafe during breastfeeding; use contraception during treatment, Renal impairment (dose adjust) |




Reviews
There are no reviews yet.